The two phases of cellular water are vital to life and all bodily functions that support it. Current higher-scale medical educational programs still teach a single phase of the water model of cellular biology. We learn in basic science class that water has three phases: liquid, solid, and gas, but for over a hundred years, science has known that the fourth phase of water exists in certain circumstances. In cellular physiology class (cellulr physiology= the study of the functions of cells), we learned that cells are like tiny bags made up of a double layer called the lipid bilayer, which holds water and has floating parts like organelles (basically the organs of a cell), and the nucleus (the core of the cell) moving around inside of it. The cell's shape is held together by a network of proteins called the cytoskeleton, which acts as a support frame in the cell. The inside of the cell is controlled by special proteins in the cell membrane called pumps and channels. Pumps push certain molecules out of the cell, while channels let others flow in. This simplistic model, on the surface, makes a lot of sense. As we dive deeper, however, issues begin to arise. For example, keeping the right balance of electrolytes inside the cell using pumps would take a lot more energy than seemingly possible. We know that cells do this, but our current model does not explain how. Energy is consistently confusing when we also look at how things move around inside the cell. Cells use a few methods such as the use of vesicles, cytoskeleton "tracks", endocytosis, and exocytosis to move materials like proteins and molecules around, even in the cell's liquid environment.
Scientists studying the theory of water's fourth phase have helped to explain the roadblocks of understanding cell functionality. Their idea is called "phase transition cellular mechanics". This idea suggests that cells can use the shift of water between its liquid form and a special fourth phase to carry out tasks inside the cell while using significantly less energy.
What is the Fourth Phase of Water
Over a hundred years ago, scientists made a discovery about the nature of water that took years to explain. When they dropped a hydrophilic (water-attracting) particle like Nafion tubing in salt water, a zone of pure water, which they later called the exclusion zone would form. The exclusion zone pushed out salt so that sodium and chloride ions could not enter. The debate started as to why this occurred. One side believed that it was created by water molecules lining up and stacking neatly like bricks The other side argued that the size of the exclusion zone was too large to maintain such a structure. The analogy would be stacking bricks as high as 3 football fields, it would not maintain itself. As we can see mainstream science still holds on to the brick theory of the exclusion zone only recognizing 3 phases of water.
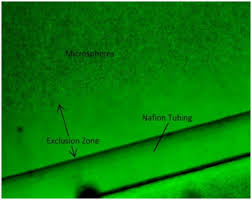
The research into the fourth phase of water has revealed many surprises along the way, carving out a new model of cellular physiology not applied by current academics in the medical field. One of the first findings about the exclusion zone was the charge separation. The exclusion zone was very negatively charged, and the zone between it along with a lot of liquid water was very positively charged.
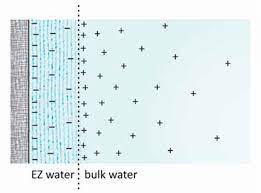
Scientists discovered that if they put an electrode in exclusion zone (EZ) water, and one at the edge, a small electric current flowed. This even happened in pure water which was surprising because usually separating electric charges like this takes energy. For example, our cells use a lot of energy to keep charged separated, but with EZ water, it forms on its own by touching surfaces that attract water which doesn't require energy. Since many parts of our body attract water, this could be important. Scientists don't fully understand how this works currently as something seems to be missing.
As time went on and technology improved, they were able to see what EZ water was composed of. The water molecules would link up in a hexagonal structure as not H2O but H3O2. The honeycomb structure stacked on top of each other made a stable structure that the brick stacking theory could not reconcile. EZ water soon became known as structured water. They still understood it existed, but again, how was the H3O2 being assembled? It would require a reaction that would need energy input.
By accident, researchers discovered the energy source driving the formation of the EZ water. Dr. Gerald Pollack was studying exclusion zone water in his lab, and a student found that shining light on a sample would make the exclusion zone larger. This was the answer to the energy input question, but was it the light or the heat of the light that was causing this to occur? They later found that it was near-infrared wavelengths of light that were exciting the electrons in the water to drive the reaction of H3O2 formation around the hydrophilic surface. Near-infrared energy is everywhere. The sun emits it, and the earth, plants, and creatures absorb and reemit it. Fire emits near infrared and far infrared, the latter being responsible for the warming effects of fire. Hydrothermal vents on the ocean floor emit near-infrared energy. In any place on Earth, water is exposed to hydrophilic substances, and water will structure around them.
Structured Water And Cellular Physiology
Cells have a membrane consisting of a hydrophobic lipid bilayer (discourages structuring of water) with many hydrophilic proteins (encourage structuring of water) that span across the membrane. Inside the cell, along the membrane, proteins form a lattice network around the entire cell. Within the cell, another protein lattice joins all of the organelles and the nucleus together. This network of proteins is encased with structured water, so much so that there is very little unstructured water within a cell. The inside of the cell is a gelatinous matrix. Think of Jello, the consistency is formed by the structuring of water around the collagen in the Jello. Our cells are similar to Jello on the inside.
Our cells use the energy stored in water to do work, and they do so by controlling how water is arranged around proteins. Water can shift between 2 forms: loose and unstructured, and organized and structured. These changes are called phase transitions, and they happen when the charge of the nearby protein changes. Cells use a molecule called ATP to make this happen. ATP molecules act as batteries for cells as they are the cell's main energy source. ATP sticks tightly to proteins and changes their shape. While the ATP is attached, the protein becomes stretched and attracts more water, causing the water around it to form a structured layer. When ATP is fully broken down and used, the protein's shape changes again and the structured water breaks apart. This sudden change releases energy, and that energy helps move ions like sodium, potassium, calcium, and chloride quickly through the cell. It also fuels important actions like muscle contraction, nerve signals, DNA copying, making proteins, and moving materials in and out of cells.

Should I Drink Structured Water?
Structured water is the new fad, making people believe that structured water will increase their energy and health. It sounds good, but what are you getting out of it? If you drink water and are exposed to infrared energy, your cells will structure it naturally. So, does it need to be structured before you drink it? All water in a glass will be structured at the surface because it is exposed to charged particles in the air (hydrophilic) and infrared energy. Clouds are small droplets of structured water around dusk, just as raindrops are. The surface tension of water is due to water structuring, which enables lizards and water bugs to walk on water.

What we should do is drink water as nature intended it. Natural water would not be fluoridated, chlorinated, and stored in a dark water tower delivered in copper pipes. It would not be sitting in a toxic plastic bottle to stagnate. Water would be exposed to sunlight and moving in nature. I think the best economical way to drink healthy water is home reverse osmosis filtration or distilling water. To get pure water, you want only H20 with nothing else in it. We recommend that when installing reverse osmosis units, to bypass the re-mineralization stage. Under the sink, units can be purchased for $300-$500 and are easy to maintain. A stovetop water distiller costs $100-$200. This is what I recommend. Also buy a couple of 5-gallon glass carboys to keep finished water, then buy a dispenser. Start with pure H2O then add a small amount of Himalayan or plain sea salt to add ions in a clear glass cup. Stir it up well to add that motion to the water and mix in the salt. Let the water then get exposed to direct sunlight or if you have access to a red/near-infrared light that would work. The light will not structure the water to any significant degree but the water will absorb the photon energy exciting the electrons within the ions and water which will then transfer to your body when you drink it.
